õ » : 网站设计/优化推广
风格: 网站设计/优化推广
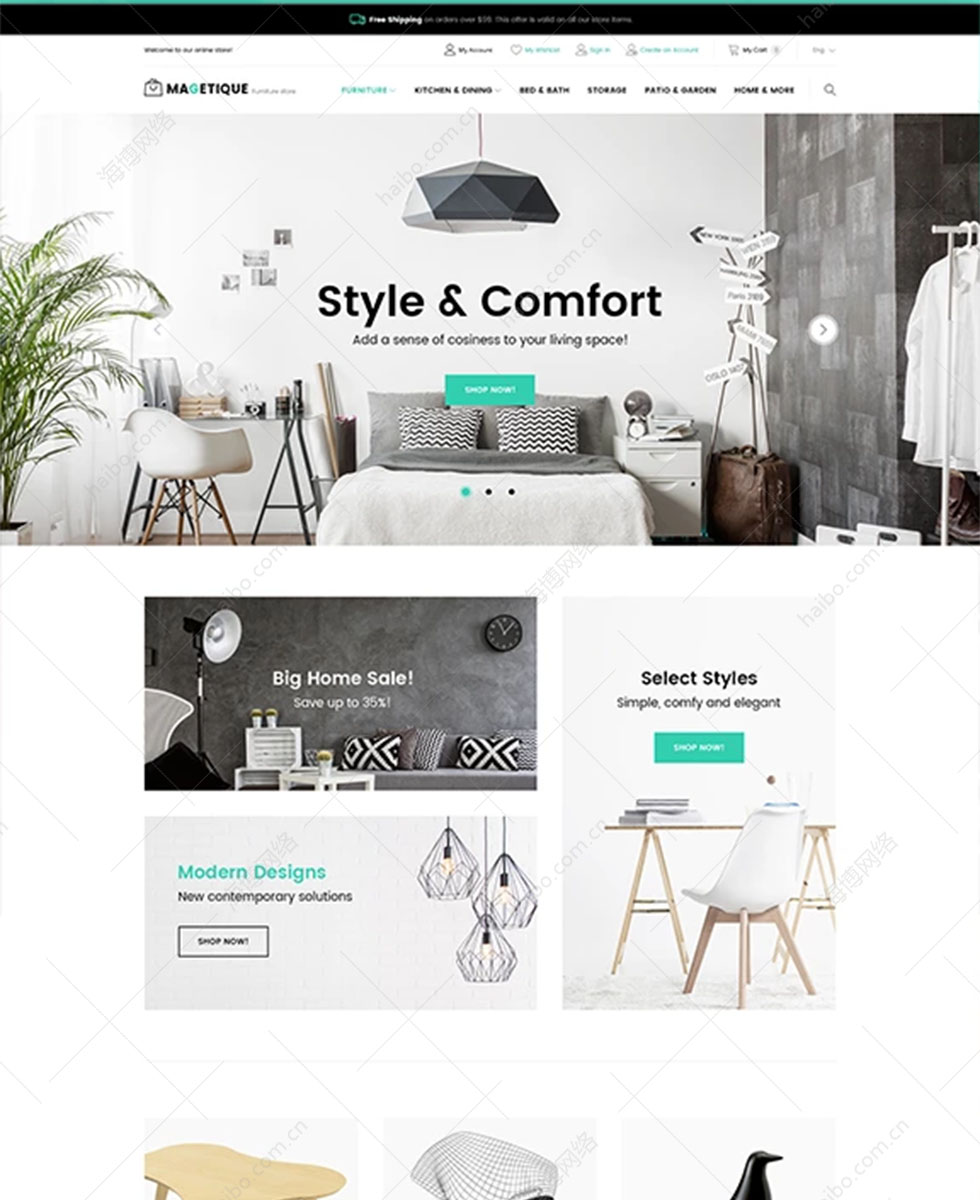
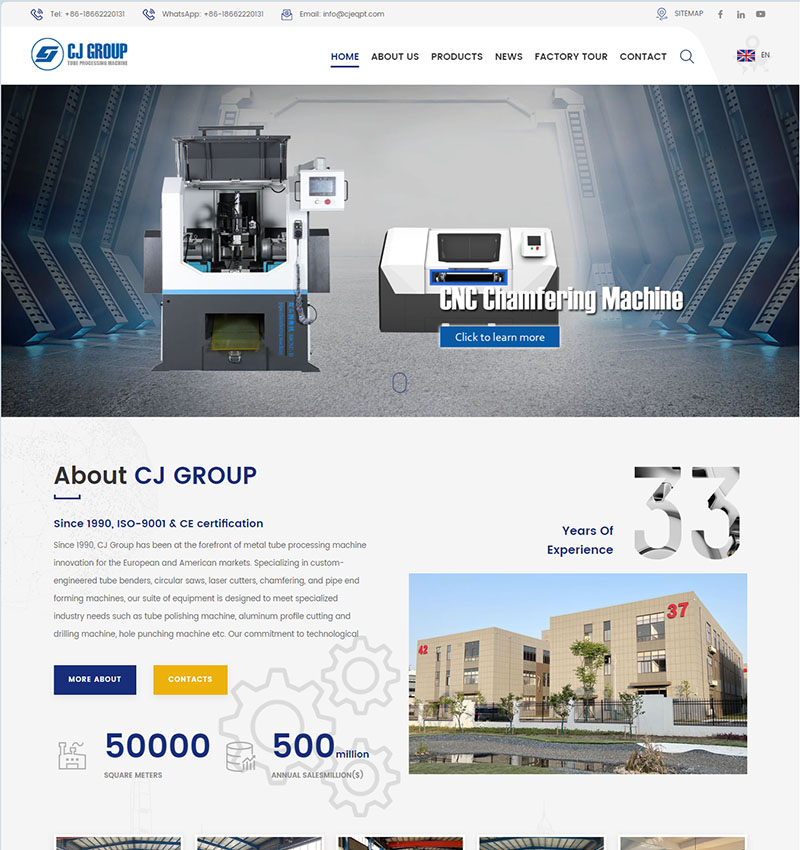
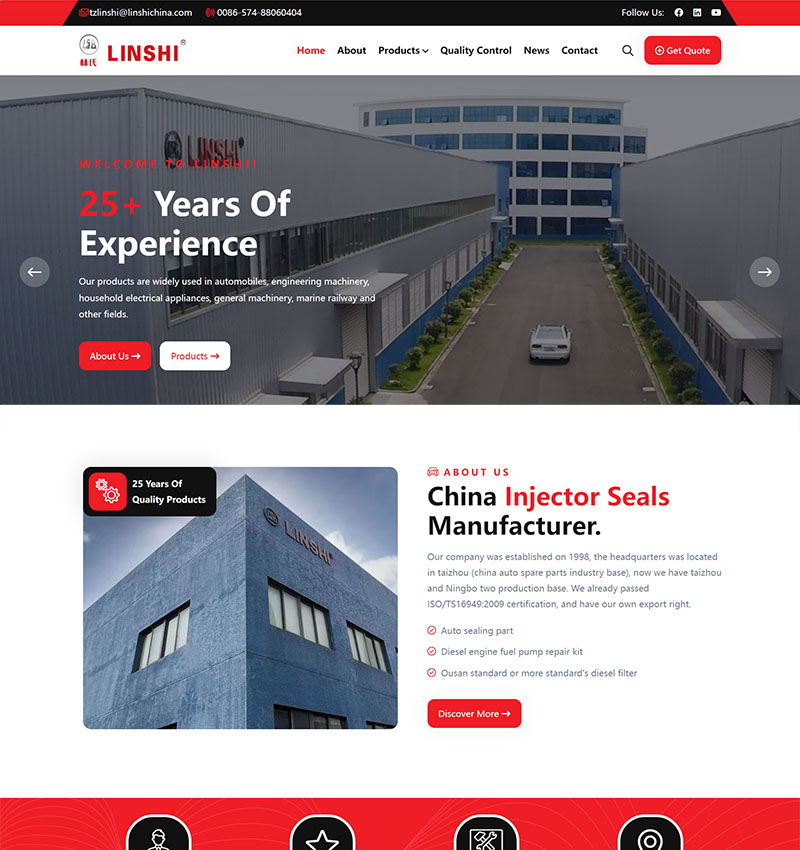
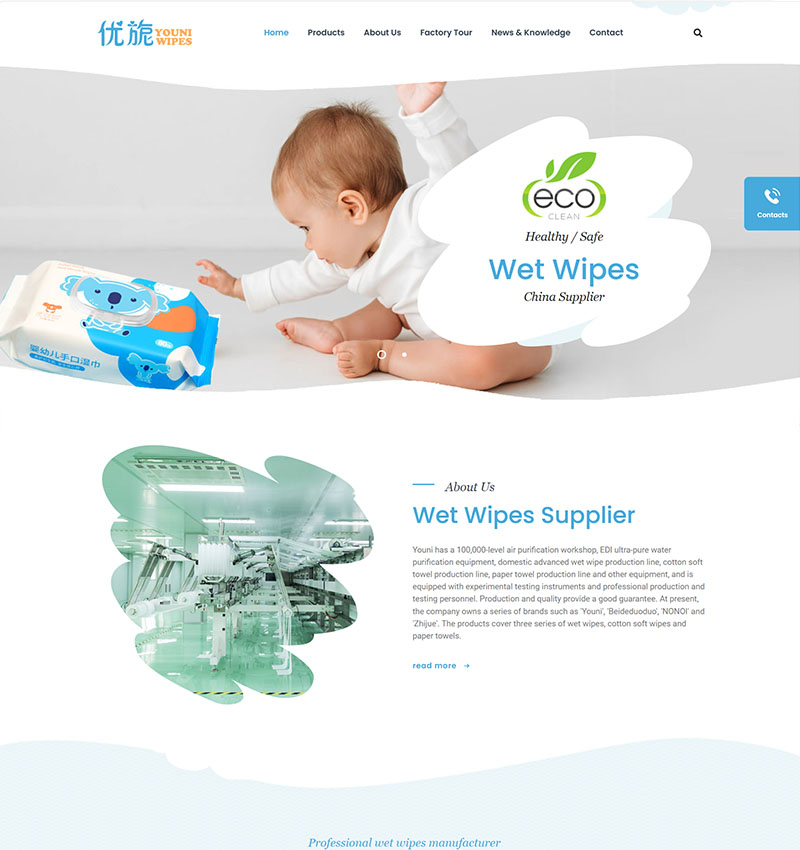
整站预览H5响应式定制设计,26种语言/SEO营销型网站
H5/响应式/营销型企业网站, 26种语言扩展,覆盖: 英文、法语、德语、西班牙语、葡萄牙语、俄语、阿拉伯语、波斯语、日语、韩语(可替换语言:马来语、泰语、越南语、菲律宾语、意大利语、荷兰语、波兰语、挪威语、芬兰语、丹麦语)
联系QQ:1817609900海博网络-为企业构建通达全球的电子商务
十五年专业服务,一站式,全网通,营销型外贸网站建设,SEO搜索引擎优化,响应式,多语言,跨境电商网站建设,外贸网站制作,外贸网站推广,外贸建站,自助建站